Ultrasound for Critical Care Physicians: Hungry Heart
 Thursday, January 14, 2016 at 8:00AM
Thursday, January 14, 2016 at 8:00AM A 31-year-old incarcerated man with a past medical history of intravenous drug use and hepatitis C, presented with a one week history of dry, non-productive cough, orthopnea and exertional dyspnea. He denied current intravenous drug use, and endorsed that the last time he used was before he was incarcerated over 3 years ago, his last tattoo was in prison, 6 months prior. He was found to have an oxygen saturation of 77% on room air, fever of 40º C, heart rate of 114 bpm, and blood pressure of 80/50 mmHg. The patient had a leukocytosis of 14 x109/L, and a chest x-ray demonstrating patchy airspace disease. Blood cultures were sent and he was treated with antibiotics and vasopressors for septic shock. The patient was intubated for acute hypoxemic respiratory failure secondary to multifocal pneumonia. A bedside transthoracic echocardiogram was performed.
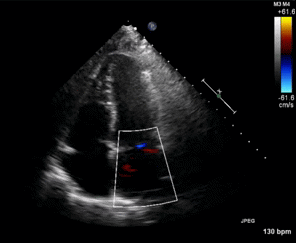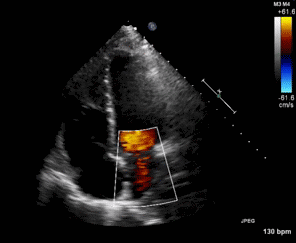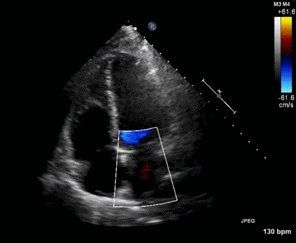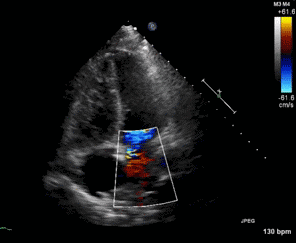
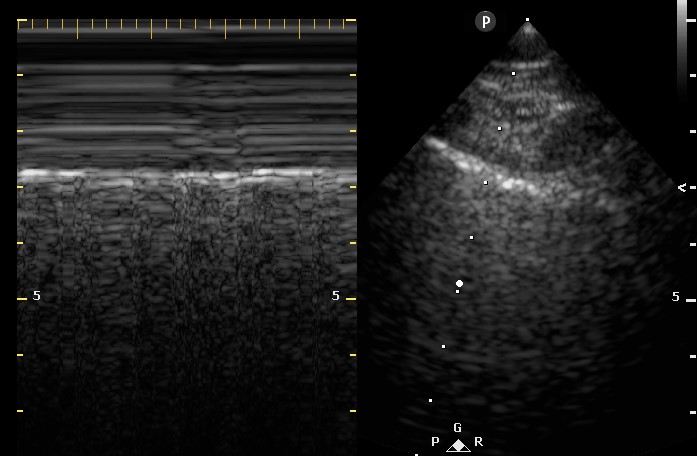
Figure 1. Apical four chamber view echocardiogram with color Doppler over the mitral valve.
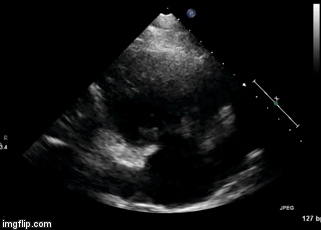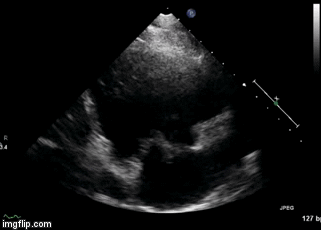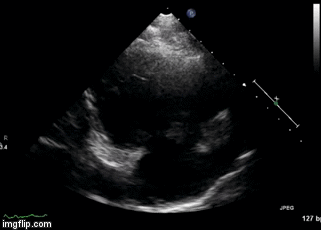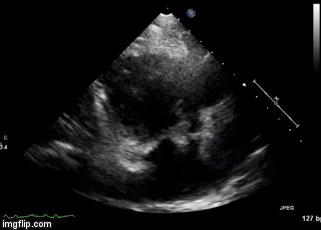
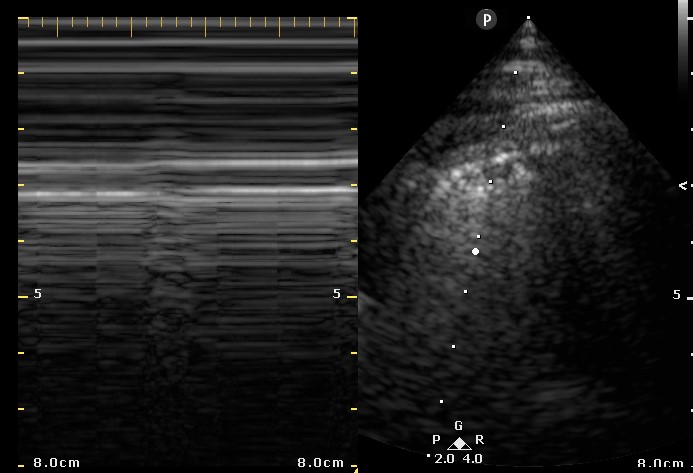
Figure 2. Right Ventricular (RV) inflow view echocardiogram from same patient What is the likely diagnosis supported by the echocardiogram? (Click on the correct answer for an explanation)
Cite as: Villalobos N, Stoltze K, Azeem M. Ultrasound for critical care physicians: hungry heart. Southwest J Pulm Crit Care. 2016;12(1):24-7. doi: http://dx.doi.org/10.13175/swjpcc007-16 PDF