Refractory Cardiogenic Shock
 Monday, October 21, 2013 at 10:10AM
Monday, October 21, 2013 at 10:10AM Bhupinder Natt, MD (bnatt@deptofmed.arizona.edu)
Tauseef Afaq Siddiqi, MD (tsiddiqi@deptofmed.arizona.edu)
Jarrod Mosier, MD (jmosier@aemrc.arizona.edu)
Yuval Raz, MD (yraz@deptofmed.arizona.edu)
Department of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, University of Arizona, Tucson, AZ
Abstract
We present a case of refractory cardiogenic shock secondary to myxedema crises that was treated successfully with thyroid hormone replacement.
Case Presentation
History of Present Illness
A 62-year-old woman with past medical history significant only for hypothyroidism was seen in the emergency department (ED) after a fall at home. On further evaluation, the patient’s husband reported history consistent with deteriorating mental function as evident by speech alteration, inability to operate home appliances like the coffeemaker and gradually worsening generalized weakness. She was recently seen at an outside hospital the week previous to this admission and given a prescription for furosemide for pedal edema.
ED evaluation showed her to be lethargic, disoriented with progressively deteriorating mentation, with hypothermia and bradycardia. Her progressive decline in mentation led to endotracheal intubation for airway protection. The patient was started on vasopressors due to post-intubation hypotension and transferred to the Medical Intensive Care Unit (MICU).
Vitals and Physical Exam
Vitals upon presentation to ED: Temperature 29o Celsius; Heart Rate 42 bpm; Blood Pressuren107/79 mm Hg; Respiratory Rate 14/min; SpO2 97 % on Room Air.
Vitals upon arrival in MICU: Temperature of 29o Celsius; Heart Rate 43 bpm; Blood Pressure 111/57 mmHg; Respiratory Rate of 10/min; SpO2 100% on mechanical ventilation;
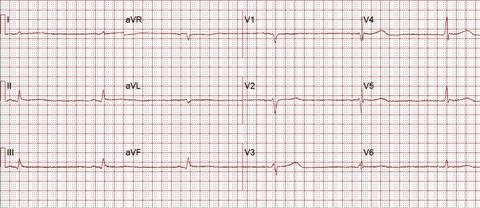
Neurologically, she was sedated with no obvious localizing signs. Skin exam revealed a sutured scalp lesion and scattered abrasion on arms. Cardiopulmonary exam was significant only for bradycardia, which was determined to be sinus rhythm by the electrocardiogram (EKG). Other system examination was significant only for 2+ non-pitting edema of upper and lower extremities.
Laboratory and Radiology Data
CBC: White Cell Count 8.4x1000/microL; Hemoglobin 9.5 g/dL; MCV 100 fL; Platelets 82x1000/microL.
Electrolytes and Metabolic Panel: Sodium 134 mMol/L; Potassium 4.2 mMol/L; Chloride 97 mMol/L; Bicarbonate 26 mMol/L; Urea Nitrogen 38 mg/dL; Creatinine 0.9 mg/dL. Glucose 78 mg/dL; Calcium 9.6 mg/dL.
Total Protein 6.3 g/dL; Albumin 3.7 g/dL; Bilirubin 0.7 mg/dL; Alkaline Phosphate 104 IU/L; ALT 72 IU/L; AST 158 IU/L; TSH 36.1 uIU/mL; Thyroxine 0.8 ng/dL; Free T3 1.2 pg/mL (Normal 2.4-4.2 pg/mL); Cortisol 20.3 mcg/dL.
ABG: pH 7.52; PaCO2 34; PaO2 166; HCO3 28; SaO2 97.6 % on 50% FiO2.
Urine Analysis: Negative.
Urine Drug Screen: Negative.
Chest X-Ray: Emphysematous changes with no acute cardiopulmonary process.
Non-contrast CT of the head and cervical spine: Unremarkable except scalp hematoma.
EKG at Presentation
MICU Course
After initial stabilization of blood pressure in ED; receiving four liters of intravenous fluids; and norepinephrine infusion at increasing doses, she was transferred to MICU. She required two more liters of crystalloids, vasopressin and increasing doses of norepinephrine, epinephrine, phenylephrine and dobutamine infusions. Corticosteroid was added for refractory shock. She also received first dose of broad-spectrum antimicrobials in ED.
Two hours after arrival to the MICU, seven hours post presentation to the ED; a Swan-Ganz pulmonary artery catheter was placed. The values obtained from the Swan-Ganz catheter are reported in table 1.
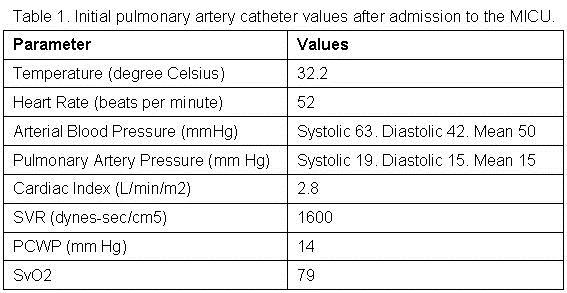
These values were obtained seven hours into admission, while the patient was being actively rewarmed, treated with a total of six liters (L) of crystalloid solutions and on norepinephrine at 13 mcg/min and dobutamine at 5 mcg/kg/min.
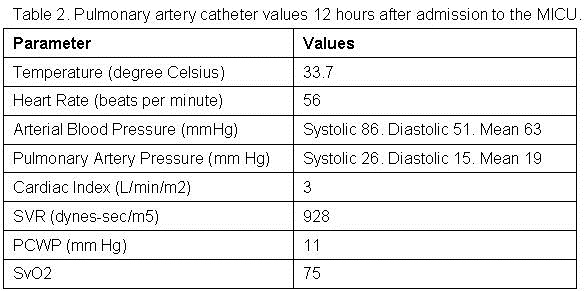
The patient remained hypotensive and sequentially, within hours, multiple vasopressors and inotropic agents were required. Table 2 reports the vitals and values off the Swan-Ganz catheter 12 hours after initial placement, when the patient is being actively rewarmed, treated for close to 18 hours and while receiving intravenous epinephrine at 3 mcg/min, norepinephrine at 30 mcg/min, vasopressin a 0.03 units/min, phenylephrine at 150 mcg/min and dobutamine at 20 mcg/min.
A formal 2-D echocardiogram was obtained which showed a global left ventricular dysfunction and hypokinesis with reduced ejection fraction of 35% and mildly reduced right ventricular dysfunction. No major valvular or pericardial pathology was noticed. Given the patient’s history and clinical features of hypothyroidism, a diagnosis of myxedema coma was made and treatment started with levothyroxine 125 mcg a day intravenously. Corticosteroids were discontinued due to concern of blockage of peripheral conversion of T4 to T3. T3 was unavailable in our city initially but was subsequently obtained and prescribed to our patient at 25 mcg of liothyronine three times a day. She responded very well to the treatment and was successfully weaned off all vasopressor support, liberated from mechanical ventilation on the fourth day and transferred out of the MICU on day five of admission. Limited repeat echocardiogram prior to transfer out of the MICU showed an ejection fraction of 65%.
Cultures, lumbar puncture and all imaging including brain MRI obtained during admission and MICU stay remained negative. A potential precipitating factor was not found and all empiric treatments were gradually withdrawn prior to the transfer out of MICU.
Retrospective history taking revealed patients’ non-compliance with levothyroxine treatment for the past 2 years. She had been given a dose of levothyroxine while she was admitted at the outside hospital the week previously leading to her relatively normal T4 level, suggesting inhibited peripheral conversion to T3 and thus the limited effect of intravenous levothyroxine initially. Surprisingly, despite the ordeal, she continued to refuse to take the medication once extubated and transferred to Medicine.
Discussion
A myxedema crisis is an extreme and life-threatening form of hypothyroidism that requires prompt diagnosis and treatment. Mortality rate has been reported as high as 60% despite appropriate diagnosis and management (1,2). Infections and discontinuation of thyroid supplements are the major precipitating factors. Other precipitants include hypothermia, gastrointestinal bleeding, congestive heart failure, cerebrovascular accident, metabolic disturbance and sedative drugs (3).
Myxedema crises manifests by involving multiple organ systems. Hypothermia can be profound and is usually the first sign of myxedema coma. Neuropsychiatric manifestations include confusion, lethargy, coma, psychosis (myxedema madness), and seizures. Respiratory depression is common likely due to depressed hypoxic and hypercapnic respiratory drive. Edema (myxedema) involving upper airways contributes to acute respiratory failure. Reduced intestinal motility leads to constipation, paralytic ileus and mega colon. Urine retention from bladder atony can be seen. Reduced glomerular filtration and reduced water excretion leads to hyponatremia, a consistent finding in myxedema crises.
Cardiovascular effects of myxedema crises include conduction abnormalities, reduced contractility, cardiomegaly and pericardial effusion. Sinus bradycardia, low voltage complexes, bundle branch blocks, complete heart blocks, and nonspecific ST-T changes in electrocardiogram have been reported. Prolonged QT interval and polymorphic ventricular tachycardia has been described as well (4). Depressed cardiac contractility leads to low stroke volume and cardiac output. Animal studies have documented that hypothyroidism leads to cardiomegaly from cardiac atrophy, impaired myocardial blood flow and loss of arterioles resulting in severe systolic dysfunction (5).
These cardiogenic effects of hypothyroid state leading to depressed ionotropy and chronotropy with compensatory vasoconstriction are believed to be a result of low intracellular T3. The hypothyroid heart attempts to perform by better coupling of ATP to contractile events until a precipitating event disrupts this fine balance (6). This resultant de-compensation leads to hypotension and cardiogenic shock that may not respond to vasopressors alone until thyroid hormone replacement is given.
Treatment usually involves intensive care admission with cardiopulmonary support, aggressive fluid and electrolyte management, treatment of underlying and precipitating factors and thyroid hormone replacement. Intravenous T4 is the most commonly used preparation as intestinal absorption may not be reliable. In severe illness, the conversion of T4 to T3 by 5’-monodeiodinase is impaired. Intravenous T3 preparation should be used. Improved cardiac function is reported in 24 hours when T3 replacement is used and it may take up to a week to notice beneficial effects with T4 replacement alone (7).
References
- Mathew V, Misgar RA, Ghosh S, Mukhopadhyay P, Roychowdhury P, Pandit K, Mukhopadhyay S, Chowdhury S. Myxedema Coma: a new look into an old crisis. J Thyroid Res. 2011:493462.[CrossRef] [PubMed]
- Wartofsky L, Myxedema Coma, Endocrinol Metab Clin N Am. 2006; 35(4): 687-98. [CrossRef][PubMed]
- Brent GA, Davies TF. Hypothyroidism and thyroiditis. In: Melmed S, Polonsky KS, Reed P, Kronenberg HM, eds. Williams Textbook of Endocrinology. Philadelphia, PA: WB Saunders, 2008.
- Schenck JB, Rizvi AA, Lin T. Severe primary hypothyroidism manifesting with torsades de pointes. Am J Med Sci. 2006 Mar; 331(3): 154-6. [CrossRef] [PubMed]
- Tang YD, Kuzman JA, Said S, Anderson BE, Wang X, Gerdes AM. Low thyroid function leads to cardiac atrophy with chamber dilatation, impaired myocardial blood flow, loss of arterioles, and severe systolic dysfunction. Circulation. 2005;112(20):3122-30. [CrossRef] [PubMed]
- W. M. Wiersinga. Hypothyroidism and myxedema coma. In: Jameson JL, Legroot LJ, eds. Endocrinology: Adult and Pediatric. Philadelphia, PA: Saunders Elseiever, 6th edition, 2010:1607-22.
- MacKerrow SD, Osborn LA, Levy H, Eaton RP, Economou P. Myxedema associated cardiogenic shock treated with intravenous triiodothyronine. Ann Int Med. 1992;117:1014-5. [CrossRef][PubMed]
Reference as: Natt B, Siddiqi TA, Mosier J, Raz Y. Refractory cardiogenic shock. Southwest J Pulm Crit Care. 2013;7(4):246-50. doi: http://dx.doi.org/10.13175/swjpcc098-13 PDF

Reader Comments (1)
frilawl e3d3fd1842 https://yaapoo.com/beamulpertterw