Management of Refractory Hypoxemic Respiratory Failure secondary to Diffuse Alveolar Hemorrhage with Venovenous Extracorporeal Membrane Oxygenation
 Friday, May 10, 2019 at 8:00AM
Friday, May 10, 2019 at 8:00AM Evanpaul Gill2
Mohamed A. Fayed1,2,
Elliot Ho1,2
University of California San Francisco - Fresno Medical Education Program
1Pulmonary and Critical Care Division
2Department of Internal Medicine
Fresno, CA USA
Abstract
Uncontrolled bleeding has been a relative contraindication for the use of venovenous extracorporeal membrane oxygenation (VV ECMO), but current practice is relatively institution dependent. With the recent advances in circuit technology and anticoagulation practices, the ability to manage patients with ongoing bleeding with ECMO support has increased. We report the case of a 66-year-old patient with refractory hypoxemic respiratory failure secondary to diffuse alveolar hemorrhage (DAH) from underlying anti neutrophil cytoplasmic antibody (ANCA) associated vasculitis who was successfully supported through his acute illness with VV ECMO. ECMO is often used to manage patients with refractory hypoxemic respiratory failure but the usage in the setting of DAH is less known given the risk of bleeding while receiving anticoagulation. Our patient was successfully managed without anticoagulation during his initial ECMO course and his respiratory failure rapidly improved after cannulation. Once managed through the acute phase of his illness and treatment started for his underlying disease process, anticoagulation was started. After being de-cannulated from ECMO and a 3 week stay in the acute rehabilitation unit, our patient was discharged home with complete recovery from his illness. We highlight that patients with refractory hypoxemic respiratory failure and suspicion of DAH as an etiology, ECMO without anticoagulation should be considered as supportive salvage therapy until the underlying process can be treated.
Case Presentation
A 66-year-old man presented with cough, fever, and dyspnea for 1 week. Upon presentation he was found to be in hypoxemic respiratory failure with bilateral pulmonary infiltrates on chest x ray (Figure 1) and positive testing for Influenza A.
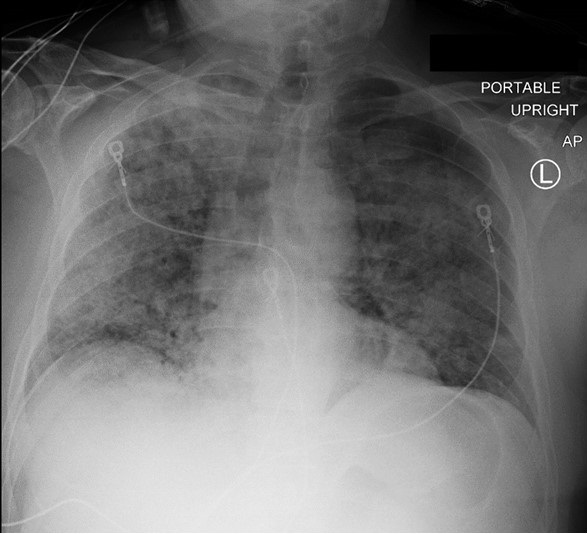
Figure 1. Portable AP of chest on initial presentation showing bilateral infiltrates more prominent on the right.
He had an elevated creatinine of 8.1 mg/dl and an acute anemia with a hemoglobin of 7.4 g/dl during the initial work up. He was intubated on hospital day one and transferred to our center for a higher level of care early morning on hospital day two. He developed refractory hypoxemic respiratory failure despite maximum ventilator support as well as standard acute respiratory distress syndrome (ARDS) treatment including neuromuscular blockade. Prone positioning was not possible secondary to hemodynamic instability during the initial treatment plan. Infectious and autoimmune work up was sent. A thoracic CT scan showed extensive bilateral consolidation (Figure 2).
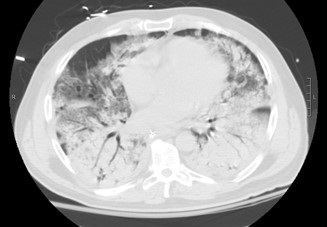
Figure 2. A representative image from the thoracic CT scan showing extensive bilateral consolidation.
At this point a decision was made to apply venovenous double lumen (VVDL) ECMO support as a supportive salvage therapy pending further evaluation into the etiology of his respiratory failure and ARDS. Etiologies at this point included severe influenza infection and DAH from an underlying vasculitis. Anticoagulation with heparin was not initiated given the significant anemia requiring multiple blood transfusions at that point. BUN was elevated, but no other signs of acute gastrointestinal bleeding were identified. Given the underlying renal failure, continuous renal replacement therapy (CRRT) was started on hospital day 2 with citrate used as the anticoagulant. After initiation of ECMO, he improved significantly in the next 72 hours, however, he developed bleeding from the endotracheal tube on day 4. Bronchoscopy was subsequently performed and showed bloody secretions throughout the respiratory bronchial tree, consistent with DAH. His ECMO course had been unremarkable with no thrombotic complications requiring changing of the circuit. Target flows were achieved with a Cardiohelp centrifugal pump and his Avalon 31F double lumen catheter was without complication. On day 5, his autoimmune panel showed a positive ANCA, with myeloperoxidase elevated at 82 AU/ml and serine protease elevated at 314 AU/ml. His anti-nuclear antibody (ANA) was also positive with his titer at 1:2,560. After rheumatology consultation, he was diagnosed with ANCA associated vasculitis with pulmonary hemorrhage and renal failure. His influenza infection was thought to be the trigger for the exacerbation of his underlying autoimmune disease. He was initiated on pulse dose steroids and plasmapheresis with significant clinical improvement and was de-cannulated from ECMO on day 8 with extubation following shortly afterward. He later had renal biopsy performed and it showed diffuse crescentic glomerulonephritis secondary to ANCA vasculitis. He was able to discontinue dialysis after requiring 8 days of CRRT and a further 3 weeks of intermittent hemodialysis. A chest x-ray showed complete clearing of the consolidation (Figure 3).
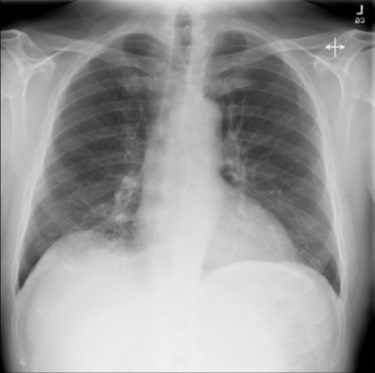
Figure 3. Chest x-ray just prior to discharge showing complete clearing of the consolidations.
He was eventually discharged home after a 3-week period in acute inpatient rehab.
Discussion
VV ECMO is increasingly being used as a viable treatment option in patients with refractory acute respiratory failure, especially in patients with underlying ARDS. The ability to allow lung protective ventilation by use of an extracorporeal circuit is of significant value in the acute phase of severe respiratory failure. The general principle of VV ECMO involves removing deoxygenated blood from a venous catheter and passing it through a closed circuit which is comprised of a centrifugal pump and membrane oxygenator (1). This membrane oxygenator takes over the function of the diseased lungs and allows gas exchange to occur, mainly oxygenating the blood and removing carbon dioxide.1 This blood is then returned into the venous circulation and eventually makes it to the systemic circulation to oxygenate the tissues.
Given that native blood is being passed through an artificial circuit, the risk for thromboembolism is thought to be relatively high. The pathophysiology behind this risk stems from contact of blood components with the artificial surface of the ECMO circuit (2). Proteins found in blood, mainly albumin and fibrinogen, will stick to the artificial surface (2). This results in other blood components congregating, which leads to the formation of a protein layer that servers as an anchor for platelet activation and the formation of insoluble fibrin clots (2). Given the risk of thromboembolism, Extracorporeal Life Support (ELSO) guidelines recommend routine anticoagulation for patients undergoing extracorporeal support (3).
The major complications regarding anticoagulation in the setting of ECMO is bleeding (4). The risk generally comes from acquired thrombocytopenia and anticoagulation (2). ELSO has guidelines regarding management of anticoagulation in VV ECMO but the current practice is relatively institution dependent. This was highlighted in a systematic review done by Sklar and colleagues (4) that investigated many different approaches to anticoagulation for patients on VV ECMO. The main anticoagulant used in those studies was unfractionated heparin and the means to measure its effect was activated clotting time (ACT) and partial thromboplastin time (PTT). They concluded that currently there is no high-quality data that can be employed in decision making regarding anticoagulation for patient’s on VV ECMO support for respiratory failure and that randomized controlled trials are needed for high quality evidence (4). Our own institution’s protocol uses unfractionated Heparin for anticoagulation with a PTT goal of 60-80.
The traditional risk of anticoagulation with ECMO has improved as the component technology of the ECMO circuit has progressed (2). Development of heparin coated inner tubing along with shorter circuit lengths are recent strategies that have been employed to help decrease the amount of thrombotic complications (2). ECLS guidelines state that patients can be managed without anticoagulation if bleeding cannot be controlled with other measures and that the use of high flow rates is recommended to help prevent thrombotic complications (3). The strategies mentioned above are non-chemical ways of preventing thrombosis and could potentially allow management of VV ECMO patients without anticoagulation for the initial period. This was demonstrated in a case report done by Muellenbach and colleagues (5). In their case series, they describe three cases of trauma patients with intracranial bleeding and severe ARDS refractory to conventional mechanical ventilation that were managed with VV ECMO without systemic anticoagulation for a prolonged time period. In their situation, anticoagulation could not be given secondary to severe traumatic brain injury (TBI) and intracranial bleeding (5). They stated that because newer circuits are completely coated by heparin and because circuit lengths have been shortened by specialized diagonal pumps and oxygenators, systemic anticoagulation can be reduced (5)
VV ECLS, as mentioned above, is commonly used in acute respiratory failure but use of VVECLS in DAH is a less known use due to the risk of anticoagulation in this clinical setting. Per ECLS guidelines, one of the relative contraindications for initiation of ECLS is risk of systemic bleeding from anticoagulation (3), and patients with DAH definitely fit this risk profile. But as mentioned above; with improving shortened ECMO circuits, use of heparin coated tubing, and high flow rates, the ability to initially manage patients without systemic anticoagulation until they are stabilized is very important in clinical settings such as our patient with DAH. Many case reports have been published that highlight the successful management of acute respiratory failure due to DAH with VV ECMO (6,7). Our patient was initially managed without systemic anticoagulation and required multiple blood transfusions given the significant bleeding appreciated from the endotracheal tube. Once the diagnosis of ANCA related DAH was made and the appropriate treatment initiated, bleeding significantly decreased and the patient was able to be started on anticoagulation. This highlights that patients with suspicion of DAH as an etiology of respiratory failure not be excluded from consideration VV ECMO as supportive salvage therapy given the potential for great clinical outcome if managed through the acute phase of bleeding.
References
- Brodie D, Bacchetta M. Extracorporeal membrane oxygenation for ARDS in adults. N Engl J Med. 2011 Nov 17;365(20):1905-14. [CrossRef] [PubMed]
- Mulder M, Fawzy I, Lance MD. ECMO and anticoagulation: a comprehensive review. Neth J Crit Care. 2018;26:6-13.
- Brogan TV, Lequier L, Lorusso R, MacLaren G, Peek G. Extracorporeal Life Support: The ELSO Red Book. Fifth Edition. Extracorporeal Life Support Organization; 2017. Thomas V. Brogan and Laurance Lequier (eds).
- Sklar MC, Sy E, Lequier L, Fan E, Kanji HD. Anticoagulation practices during venovenous extracorporeal membrane oxygenation for respiratory failure. A systematic review. Ann Am Thorac Soc. 2016 Dec;13(12):2242-50. [CrossRef] [PubMed]
- Muellenbach RM, Kredel M, Kunze E, Kranke P, Kuestermann J, Brack A, Gorski A, Wunder C, Roewer N, Wurmb T. Prolonged heparin-free extracorporeal membrane oxygenation in multiple injured acute respiratory distress syndrome patients with traumatic brain injury. J Trauma Acute Care Surg. 2012 May;72(5):1444-7. [CrossRef] [PubMed]
- Abrams D, Agerstrand CL, Biscotti M, Burkart KM, Bacchetta M, Brodie D. Extracorporeal membrane oxygenation in the management of diffuse alveolar hemorrhage. ASAIO J. 2015 Mar-Apr;61(2):216-8. [CrossRef] [PubMed]
- Patel JJ, Lipchik RJ. Systemic lupus-induced diffuse alveolar hemorrhage treated with extracorporeal membrane oxygenation: a case report and review of the literature. J Intensive Care Med. 2014 Mar-Apr;29(2):104-9. [CrossRef] [PubMed]
Cite as: Gill E, Fayed MA, Ho E. Management of refractory hypoxemic respiratory failure secondary to diffuse alveolar hemorrhage with venovenous extracorporeal membrane oxygenation. Southwest J Pulm Crit Care. 2019;18(5):135-40. doi: https://doi.org/10.13175/swjpcc007-19 PDF

Reader Comments